Laboratory Integration & Management Solutions
Empowering Labs Through Seamless Technology Integration
AtLab LLC is a U.S.-based technology service provider specializing in Laboratory Information Management Systems (LIMS). We enable laboratories to streamline their operations, enhance data accuracy, and boost productivity through robust, customized software solutions.
With a commitment to quality and around-the-clock support, we deliver world-class lab automation systems tailored to clinical, pharmaceutical, and research sectors.
Global Collaboration, Local Commitment
AtLab success is built on strategic international partnerships. Our software development teams are based in:
- USA
- Australia
- Pakistan
These expert teams work closely with us to develop, test, and deploy cutting-edge LIMS software, while we manage direct client engagement and 24/7 technical support from our U.S. headquarters.

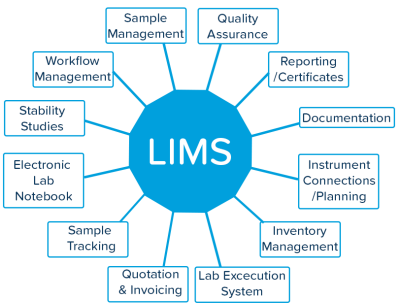
Our Core Offering LIMS (Laboratory Information Management System)
A LIMS connects laboratory instruments, software platforms, and data repositories into one integrated system. This enables:
- Automation of lab workflows
- Error-free data capture & reporting
- Fast and reliable test result delivery
- Regulatory compliance & audit readiness
- Centralized data storage and access
Our solutions are customized for a wide range of industries, including:
- Clinical Diagnostics
- Research Laboratories
- Biotechnology & Pharmaceuticals
Whether you’re a growing lab or an enterprise-scale facility, we provide scalable LIMS solutions that adapt to your unique workflows and compliance needs.
Why Choose AtLab LLC?

We don’t offer one-size-fits-all software. Every LIMS we deliver is tailored to fit your lab’s specific instruments, processes, and reporting requirements.

Our U.S.-based support team ensures your lab operations never stop. We provide real-time assistance across time zones to maintain uptime and efficiency.

All systems are developed with a focus on HIPAA, CLIA, FDA 21 CFR Part 11, and other relevant standards.

Thanks to our global dev teams, we offer fast turnarounds on system customization, deployment, and feature enhancements.

FDA Compliance
Achieve FDA Medical Device Compliance with Confidence.
When it comes to Medical Device compliance 510(k) Exempt Does Not Mean Unregulated.
Many assume 510(k) exempt means free from oversight—but even exempt devices must meet FDA and global regulatory requirements. Overlooking a single step can lead to costly delays, import detentions, or even market removal.
efore you can sell in the U.S., the first step is getting your registration done right.
ATLab simplifies FDA regulations for medical device manufacturers with unmatched expertise. We are your trusted partner assisting in achieving compliance, avoiding costly delays, and reaching the US market faster.


Comprehensive Services:
We help companies properly comply with US FDA Medical Device requirements including:
- Registration Management & Device Listing: Handle mandatory FDA filings effortlessly.
- Official US Agent: Represent your business with US-based operational requirements
- UDI / GUDID Set-Up & Submissions: Simplify unique device identification and database submissions
- Device Classification Support: Accurately determine the FDA Class for your medical device
- eMDR Service: Streamline electronic medical device reporting
- Detention Assistance: Resolve compliance holds effectively
- ComplyHub Self-monitoring: Assure your regulatory compliance with ease
- Online Compliance Training: Empower your team with up-to-date compliance education
The Most Experienced in the Industry
With decades of expertise navigating the FDA’s regulatory landscape, we provide a proven track record of success for medical device manufacturers.
Save Time, Avoid Penalties
We ensure your registrations, listings, and filings are accurate and submitted on time, so you can focus on innovation and manufacturing.
Contact
-
ATLab LLC
7087 Spring Garden
Dr, Unit 202 Springfield, VA 22150, USA
- info@atlabllc.com
- +1 703 906 6440
Site
- Home
- About
- Products
- Services
- Contact
©2025, HelpNest. All rights reserved.

